In the first few weeks following the launch of oral semaglutide, early prescription activity answered a familiar question: Uptake was occurring and growing exponentially within a matter of weeks. But a more consequential signal emerged when looking beyond prescription counts to the patients themselves.
After analyzing NorstellaLinQ EMR and open claims data, we found that approximately three-quarters of patients initiating oral Wegovy had not previously received a GLP-1 injection. This is despite an average BMI of 35—a data point found within clinician notes in the EMR—placing them squarely within the population clinically eligible for existing injectable therapies. These were not patients newly qualifying for treatment. They were patients who had been eligible, but untreated.
In this context, early adoption reflects not only competitive dynamics within the GLP-1 class, but the activation of latent demand; clearly, early uptake was not driven solely by switching behavior. The oral formulation appears to be bringing new patients into treatment rather than simply shifting share among existing users.
This distinction matters just as much as, if not more than, early uptake volume. It suggests that route of administration, rather than lack of clinical need, may be a meaningful barrier to initiating weight loss therapy.
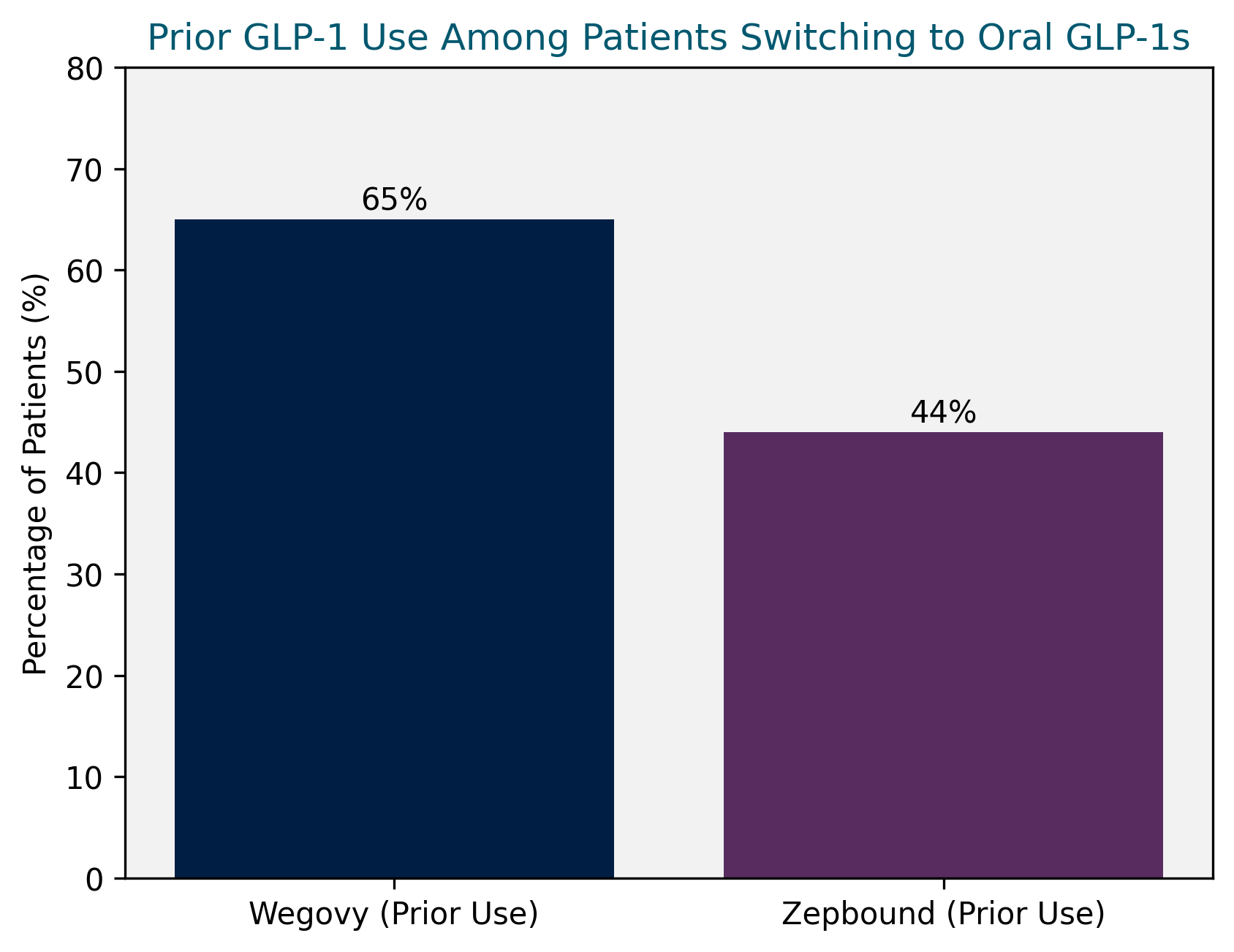
NorstellaLinQ also enabled early characterization of the roughly one-quarter of initiators who switched from prior GLP-1 therapy. Among these patients, 65% had previously used Wegovy and 44% had used Zepbound, suggesting that oral semaglutide is not simply drawing from outside the class, but is also reshaping preferences among established GLP-1 users. Notably, these patients had already demonstrated willingness to pursue pharmacologic weight loss, indicating that the oral formulation may be influencing treatment choice even among experienced users, rather than reflecting delayed entry or newly established eligibility.
If oral GLP-1s are able to engage patients who were previously unwilling or unable to initiate injectable therapy, expectations for oral uptake may need to be reconsidered. Understanding who is initiating therapy provides critical context for how the market may evolve.
Why Early Adoption Requires More Than Prescription Counts
Traditional launch tracking often relies on claims data that becomes available months after launch. While this data can confirm that prescriptions are occurring, it provides limited insight into patient characteristics, prior treatment eligibility, or the clinical rationale behind initiation.
To assess early adoption while it is still unfolding, and to interpret what that adoption actually represents, manufacturers need timely, accurate, high-resolution data that captures both the pace of uptake and the profiles of patients driving it.
As evidenced by our analysis, this is particularly true in the emerging oral GLP-1 market, where subtle differences in patient behavior and prescribing rationale can have outsized strategic implications. Early uptake alone does not fully capture what is happening. This has several important implications for manufacturers:
- Early launch benchmarks may understate success if they focus primarily on switching behavior rather than new-to-therapy initiation
- The addressable market for oral GLP-1s may be larger than previously modeled, particularly if similar barriers exist across obesity and cardiometabolic indications
- Pipeline assets following oral semaglutide may enter a more receptive market, where the viability of oral administration has already been established
What Great Looks Like: Speed and Clinical Depth at Launch
Imagine if, within weeks of launch, pharma leaders could understand the earliest signals of launch performance—including prescription activity, patient flow, site of care, and early access dynamics—by assessing near-real-time, large-scale claims and EMR data.
Approximately three-quarters of patients initiating oral Wegovy had not previously received a GLP-1 injection, according to NorstellaLinQ data
When available quickly, claims and EMR data do what claims data alone cannot. EMR data and unstructured clinical notes capture rich clinical context from patient and HCP encounters, including telehealth visits, physician rationale, and documented treatment considerations—such as why an HCP chooses a GLP-1 and why they may prefer an oral option over an injectable. Clinical notes also surface early life cycle insights, including approval outcomes and barriers to initiation, which are often invisible in claims data. Other meaningful insights include:
- Patient characteristics associated with early uptake
- Height and weight, enabling calculation of exact BMI, which is critically relevant for this market
- Clinician-documented attributes relevant to treatment selection, such as other vitals, lifestyle factors, social determinants of health, and other GLP-1 decision drivers
- HCP prescribing behavior, including identification of early adopters
- Source of business
- Differentiation between new-to-therapy patients and switchers
Rich insights like these—captured early— are necessary to understand who is being treated, why specific therapies are chosen, and whether early adoption reflects substitution within the category or expansion of the treated population. Once critical early signals are identified, pharma companies can make more informed decisions around field deployment, health system prioritization, distribution planning, and access strategy. They also help identify potential friction points, such as prior authorizations, allowing teams to respond proactively rather than waiting for lagging confirmation.
Developing a Resilient Strategy, Not a Reactive One
With additional oral GLP-1 options coming on the market, patients and physicians have more choices than ever. In this environment, launch success depends not only on generating early prescriptions, but on understanding which patients are initiating therapy and how that aligns with expectations.
Capturing these insights early requires three critical components:
- Scale: Sufficient patient volume to detect meaningful signals
- Depth: Access to clinical context not captured in claims
- Speed: Data availability within weeks, not months
Many data sources provide one or two of these elements. Bringing all three together enables early, defensible insights when timing matters most. For oral GLP-1s in particular, understanding both the pace of adoption and the profile of early adopters is essential to launch success, and to being first to insight.
As competition to understand early adoption of oral GLP-1s intensifies, manufacturers increasingly seek data partners that can deliver timely, clinically rich insights at scale. NorstellaLinQ is purpose-built to support this need, helping teams move faster, publish sooner, and act with greater confidence. Contact us to learn more.

